IEEE Boston Section
News & Announcements!
Business Manager Position Available
The Institute of Electrical and Electronics Engineers (IEEE) is looking for a Business Manager for its Boston area operations. This person should have experience in overseeing business operations, finance, marketing, social media communications, conferences, and events. For more details and to apply, click here!
IEEE Boston Section – Call for Committee Volunteers!
Upcoming Events!
- Categories
- Tags -Courses Fall 2015 -Courses-Fall-14 -Courses-Spring-15 Aerospace & Electronic Systems Antennas & Propagation Communications Computational Intelligence Computer con Consultants' Network Consumer Electronics Control Systems Courses - Spring 2016 Education el Electromagnetic Compatibility Electron Devices engineering Engineering in Medicine and Biology Societies Entrepreneurs' Network Geo-Science & Remote Sensing Industry Applications Society Life Members Magnetics Member Development Membership Development Microsystems Microwave Theory and Techniques Nuclear & Plasma Sciences PACE Photonics Power Electronics Power Energy Society re Reliability Robotics and Automation sensors council sig Signal Processing Society for Social Implications of Technology Solid State Circuits technology & management tem Women in Engineering Young Professionals
COURSE DESCRIPTION
Course Kick-off / Orientation Thursday, April 18, 6:00PM – 6:30PM.
Live Workshops: 6:00PM – 7:30PM, Thursdays, April 25, May 2, 9, 16, 23
Registration is open through the last live workshop date. Live workshops are recorded for later use.
Attendees will have access to the recorded session and exercises for two months (until July 23, 2024) after the live session ends!
Speaker: Dan Boschen
IEEE Member Fee (by April 11th): $190.00
IEEE Member Fee (after April 11th): $285.00
IEEE Non-Member Fee (by April 11th): $210.00
IEEE Non-Member Fee (after April 11th) $315.00
Decision to run/cancel course: Friday, April 12, 2024
COURSE DESCRIPTION
New Format Combining Live Workshops with Pre-recorded Video
This is a hands-on course providing pre-recorded lectures that students can watch on their own schedule and an unlimited number of times prior to live Q&A/Workshop sessions with the instructor. Ten 1.5 hour videos released 2 per week while the course is in session will be available for up to two months after the conclusion of the course.
Course Summary
This course is a fresh view of the fundamental and practical concepts of digital signal processing applicable to the design of mixed signal design with A/D conversion, digital filters, operations with the FFT, and multi-rate signal processing. This course will build an intuitive understanding of the underlying mathematics through the use of graphics, visual demonstrations, and applications in GPS and mixed signal (analog/digital) modern transceivers. This course is applicable to DSP algorithm development with a focus on meeting practical hardware development challenges in both the analog and digital domains, and not a tutorial on working with specific DSP processor hardware.
Now with Jupyter Notebooks!
This long-running IEEE Course has been updated to include Jupyter Notebooks which incorporates graphics together with Python simulation code to provide a “take-it-with-you” interactive user experience. No knowledge of Python is required but the notebooks will provide a basic framework for proceeding with further signal processing development using that tools for those that have interest in doing so.
This course will not be teaching Python, but using it for demonstration. A more detailed course on Python itself is covered in a separate IEEE Course “Python Applications for Digital Design and Signal Processing”.
Students will be encouraged but not required to load all the Python tools needed, and all set-up information for installation will be provided prior to the start of class.
Target Audience:
All engineers involved in or interested in signal processing applications. Engineers with significant experience with DSP will also appreciate this opportunity for an in-depth review of the fundamental DSP concepts from a different perspective than that given in a traditional introductory DSP course.
Benefits of Attending/ Goals of Course:
Attendees will build a stronger intuitive understanding of the fundamental signal processing concepts involved with digital filtering and mixed signal analog and digital design. With this, attendees will be able to implement more creative and efficient signal processing architectures in both the analog and digital domains. The knowledge gained from this course will have immediate practical value for any work in the signal processing field.
Topics / Schedule:
Pre-recorded lectures: (3 hours each) will be distributed Friday prior to each week’s workshop dates. Workshop/Q&A Sessions are 6 – 7:30PM on the dates listed below.
Kick-off / Orientation: Thursday, April 18, 2024
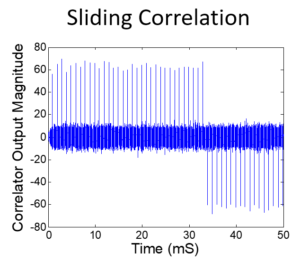
Class 1: April 25, 2024: Correlation, Fourier Transform, Laplace Transform
Class 2: May 2, 2024: Sampling and A/D Conversion, Z –transform, D/A Conversion
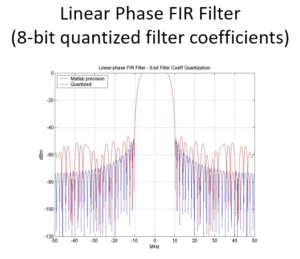
Class 3: May 9, 2024: IIR and FIR Digital filters, Direct Fourier Transform
Class 4: May 16, 2024: May Windowing, Digital Filter Design, Fixed Point vs Floating Point
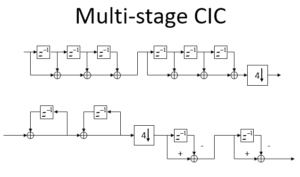
Class 5: May23, 2024: Fast Fourier Transform, Multi-rate Signal Processing, Multi-rate Filters
Speaker’s Bio:
Dan Boschen has a MS in Communications and Signal Processing from Northeastern University, with over 25 years of experience in system and hardware design for radio transceivers and modems. He has held various positions at Signal Technologies, MITRE, Airvana and Hittite Microwave designing and developing transceiver hardware from baseband to antenna for wireless communications systems. Dan is currently at Microchip (formerly Microsemi and Symmetricom) leading design efforts for advanced frequency and time solutions.
For more background information, please view Dan’s Linked-In page at: http://www.linkedin.com/in/danboschen
Registration is open through the last live workshop date. Live workshops are recorded for later use.
Consultants’ Network
Consultants’ Network: https://boston-consult.org
Free Dinner starts at 6:00 PM
Meeting starts at 7:00 PM – On-site and Zoom
Members in good standing are encouraged to attend. If you are unable to attend, then we will also be on Zoom.
Your registration confirmation email contains the Zoom details.
As a group of skilled consultants associated with IEEE Boston Section, CNET offers high value professional services for a diverse audience.
For CNET members:
- Professional interactions with peers in a supporting environment.
- General information from insiders about the condition of industry.
- Practical information exchanges about consulting practice.
- Communication improvement across multiple specialties.
- Participation in info-professional events on themes of interest.
- Opportunity to give presentations and hone speaking effectiveness.
- Credibility and visibility within the engineering and technology development community as an IEEE associated consultant.
For engineers interested in becoming consultants:
- Basic information on consulting practice requirements.
- Participation in events and idea exchanges with industry peers.
- Access to established consultants in the area for mentoring.
- Opportunities for improving professional communications skills.
For enquirers on CNET services:
- Convenient access to local consultants with multi-disciplinary expertise.
- Opportunities to enter into consulting agreements without agency fees.
- Direct, easy connection with consultants over the CNET web page.
- Dealing with consultants with multi-year experience validated by IEEE.
Join us!
Entrepreneurs’ Network
Register at https://bostonenet.org/events/funders_and_founders
The startup world can only exist with founders and funders (customers help, too). For entrepreneurs with big ideas, it can be difficult to ask for money. Where do you go to ask, how do you approach someone, and what may be most effective? In this program, you’ll hear from pros on both sides of the table.
A large segment of the population struggles to secure the funds to be successful: people of color and women. Panelists will talk about the obstacles and how to get around them.
You’ll learn to:
- Gain self-confidence to ask for funding.
- Identify potential sources to help launch your business.
- Determine what agreements may be best for the founder and funder.
- Hear from a founder who is actively seeking investors.
Event Schedule
7:00 pm Introduction, ENET Chairperson’s announcements
7:10 pm eMinute Pitch, up to 3 Startup pitches
7:25 pm Expert Panel, expert speakers on the night’s topic
8:15 pm Q & A Moderator and Audience Q & A with the speakers
8:30 pm Networking, panelists will be available afterward for responses to individual questions.
Speakers:
Dr. Cassandravictoria (Cvic) Innocent
Cvic is the General Partner of Frankenbuild Ventures — or F(v) — a MA-based VC firm investing in companies building hardtech to function in extreme environments. Her professional background blends experiences of being an investment director with her experiences in instrument development of deeptech. As such, she is very well-networked in the prototype-development communities, and the startup ecosystem in Boston and beyond. Having previously worked for NASA (2007-2009), in 2023 she was invited to give a six-seminar series of videos explaining and promoting NASA’s SBIR efforts with engineering founders and in 2024 she was listed as a Woman to Watch in VC by Amplify.
Collette Phillips – CEO and President/Chief Strategist/Visionary Colette Phillips Communications, Inc.
Colette Phillips is an extraordinary leader, an iconic cultural change catalyst, DE&I pioneer, thought leader, innovator, practical visionary, influencer, PR and marketing Maven, social commentator, philanthropist, serial social entrepreneur and uber connector. A passionate advocate for diversity and inclusion, and a driving force behind positive social change here. As the CEO of Colette Phillips Communications and the Founder of the award-winning cross-cultural Get Konnected! business networking organization. She has dedicated her life and career to empowering individuals, bridging communities, and fostering a more inclusive society and has played a pivotal role in reshaping the landscape of Boston’s business and social scenes. With her dynamic leadership at the helm of her agency, a strategic communications and public relations firm, she has spearheaded groundbreaking initiatives that have elevated the voices of underrepresented communities. She is the Founder and President of The GK Fund (A Non-Profit Social Impact Fund) that provides capital, technical and legal support, mentorship, and access to opportunities to help small BIPOC and immigrant owned businesses scale. In 2021 her company served as the lead agency for the groundbreaking multiple award-winning All Inclusive Boston Campaign in partnership with Proverb Agency and the Greater Boston Convention and Visitors Bureau to reimagine and re-energize Boston’s tourism and image. For more than three decades, Colette has been a powerful voice and effective agent of change in one of America’s oldest and most tribal cities. An immigrant Black woman from the Caribbean Island of Antigua, Boston Business Journal named her on its annual Power 50 list five times, calling her a Game-changer and a Movement Maker; Boston Magazine dubbed her “Boston’s Great Connector,” and listed multiple times on its annual Most Influential Bostonians list and she has been cited by Boston Globe for “Changing the Conversation about Diversity in Boston.” As a testament to her exceptional contributions, in 2023 she was the subject of a Harvard Business School case study for creating a DE&I ecosystem with her initiatives including the GK Fund; the groundbreaking GK! Market a digital e-commerce platform and marketplace for BIPOC and women-owned small businesses to sell their good and service as well as help corporations and nonprofit organizations create an inclusive procurement approach to their supplier diversity program; the GK! Execu-Search to connect corporate and nonprofit organizations to the Get Konnected network of robust culturally diverse talent pool for mid- level, c-suite and board positions; and the GK! Job Hub helping diverse talents find jobs with some of the best companies in Boston and beyond.
She is the recipient of numerous awards and accolades for her personal, philanthropic, and professional accomplishments including a Pinnacle Award from the Greater Boston Chamber of Commerce; Massachusetts Women’s Political Caucus’ Abigail Adams Award; The American Jewish Committee (AJC) Coexistence Award the Boston Municipal Bureau’s Shattuck City Champion Award and Boston Business Journal’s Lifetime Achievement Award for Diversity and the Boston Chapter of the Public Relations Society of America’s Lifetime Achievement Award. Her newly released book The Includers: The 7 Traits of Culturally Savvy Anti-racist Leaders has been receiving rave reviews from media and business leaders for its strategic, practical actionable insights. She holds a BS and MS from Emerson College and an Honorary Doctor of Laws from Mount Ida College.
Colette currently serves on the Advisory board of Eastern Bank; the Board of Directors for the Greater Boston Chamber of Commerce; the American Jewish Committee; The Women’s Edge and is an honorary trustee for Massachusetts General Hospital.
Ahmed Fazly – Co-founder / CEO of Modicure Therapeutics
Ahmed Fazly earned his doctoral degree in Biomedical Sciences from the Mayo Clinic College of Medicine in Rochester, MN. He began his postdoctoral journey at the University of Massachusetts Medical School in Worcester, where his research produced promising antifungal drug candidates. After teaching high school chemistry and biology at a Boston charter school, he returned to research as a postdoctoral associate in Biological Engineering at MIT, specializing in RNA modifications. His work at MIT secured STTR funding from the NIH and the Congressionally Directed Medical Research Program of the Department of Defense.
Transitioning into finance, Ahmed became a wall street sell-side analyst specializing in the rare disease therapeutics sector. His entrepreneurial drive was fostered through the Wharton School at the University of Pennsylvania’s VIP program. After contributing to commercial strategy at a biotech company in Cambridge and completing his MBA, he once again embraced entrepreneurship as the co-founder of a startup dedicated to pioneering gene editing therapies for severe genetic diseases lacking effective treatments.
Moderator – Howard Sholkin, VP ENET
Howard Sholkin has several decades of marketing communication experience across industry sectors. Since 2018, he has served as President of Newton Community Pride, a non-profit supporting a few dozen events in the city where he is a lifelong resident. In 2014, he formed Sholkin Consulting to deliver digital marketing services to technology and financial services companies. He also served on the adjunct faculty of Boston University and Lasell College for five years.
In 2003, he was hired as director of corporate communications at International Data Group (IDG) where he served Founder Pat McGovern. Sholkin later became director of communication and marketing programs for the CEO of IDG Communications, a global technology media and events subsidiary. IDG brands include Computerworld, PCWorld, Macworld, and CIO. Sholkin has held senior marketing and communication positions at technology product and services companies such as Computervision; Technology Concepts, a subsidiary of Bell Atlantic; Corporate Software; and, Digital Equipment Corp./Compaq. He was a columnist for PR News and for Personal Branding magazine, one of the first publications on social media.
Since 1990, Sholkin has been active in the Public Relations Society of America (PRSA) where he once served as president of the Boston chapter. Currently, he is co-chair of the national PRSA Investment Committee. He serves on the boards of three non-profits: Newton Community Pride, Boston Entrepreneurs Network (ENET) and Temple Shalom of Newton.
COURSE DESCRIPTION
Course Kick-off / Orientation Thursday, April 18, 6:00PM – 6:30PM.
Live Workshops: 6:00PM – 7:30PM, Thursdays, April 25, May 2, 9, 16, 23
Registration is open through the last live workshop date. Live workshops are recorded for later use.
Attendees will have access to the recorded session and exercises for two months (until July 23, 2024) after the live session ends!
Speaker: Dan Boschen
IEEE Member Fee (by April 11th): $190.00
IEEE Member Fee (after April 11th): $285.00
IEEE Non-Member Fee (by April 11th): $210.00
IEEE Non-Member Fee (after April 11th) $315.00
Decision to run/cancel course: Friday, April 12, 2024
COURSE DESCRIPTION
New Format Combining Live Workshops with Pre-recorded Video
This is a hands-on course providing pre-recorded lectures that students can watch on their own schedule and an unlimited number of times prior to live Q&A/Workshop sessions with the instructor. Ten 1.5 hour videos released 2 per week while the course is in session will be available for up to two months after the conclusion of the course.
Course Summary
This course is a fresh view of the fundamental and practical concepts of digital signal processing applicable to the design of mixed signal design with A/D conversion, digital filters, operations with the FFT, and multi-rate signal processing. This course will build an intuitive understanding of the underlying mathematics through the use of graphics, visual demonstrations, and applications in GPS and mixed signal (analog/digital) modern transceivers. This course is applicable to DSP algorithm development with a focus on meeting practical hardware development challenges in both the analog and digital domains, and not a tutorial on working with specific DSP processor hardware.
Now with Jupyter Notebooks!
This long-running IEEE Course has been updated to include Jupyter Notebooks which incorporates graphics together with Python simulation code to provide a “take-it-with-you” interactive user experience. No knowledge of Python is required but the notebooks will provide a basic framework for proceeding with further signal processing development using that tools for those that have interest in doing so.
This course will not be teaching Python, but using it for demonstration. A more detailed course on Python itself is covered in a separate IEEE Course “Python Applications for Digital Design and Signal Processing”.
Students will be encouraged but not required to load all the Python tools needed, and all set-up information for installation will be provided prior to the start of class.
Target Audience:
All engineers involved in or interested in signal processing applications. Engineers with significant experience with DSP will also appreciate this opportunity for an in-depth review of the fundamental DSP concepts from a different perspective than that given in a traditional introductory DSP course.
Benefits of Attending/ Goals of Course:
Attendees will build a stronger intuitive understanding of the fundamental signal processing concepts involved with digital filtering and mixed signal analog and digital design. With this, attendees will be able to implement more creative and efficient signal processing architectures in both the analog and digital domains. The knowledge gained from this course will have immediate practical value for any work in the signal processing field.
Topics / Schedule:
Pre-recorded lectures: (3 hours each) will be distributed Friday prior to each week’s workshop dates. Workshop/Q&A Sessions are 6 – 7:30PM on the dates listed below.
Kick-off / Orientation: Thursday, April 18, 2024
Class 1: April 25, 2024: Correlation, Fourier Transform, Laplace Transform
Class 2: May 2, 2024: Sampling and A/D Conversion, Z –transform, D/A Conversion
Class 3: May 9, 2024: IIR and FIR Digital filters, Direct Fourier Transform
Class 4: May 16, 2024: May Windowing, Digital Filter Design, Fixed Point vs Floating Point
Class 5: May23, 2024: Fast Fourier Transform, Multi-rate Signal Processing, Multi-rate Filters
Speaker’s Bio:
Dan Boschen has a MS in Communications and Signal Processing from Northeastern University, with over 25 years of experience in system and hardware design for radio transceivers and modems. He has held various positions at Signal Technologies, MITRE, Airvana and Hittite Microwave designing and developing transceiver hardware from baseband to antenna for wireless communications systems. Dan is currently at Microchip (formerly Microsemi and Symmetricom) leading design efforts for advanced frequency and time solutions.
For more background information, please view Dan’s Linked-In page at: http://www.linkedin.com/in/danboschen
Registration is open through the last live workshop date. Live workshops are recorded for later use.
IEEE Boston Section recognized for Excellence in Membership Recruitment Performance

IEEE Boston Section was founded Feb 13, 1903, and serves more than 8,500 members of the IEEE. There are 29 chapters and affinity groups covering topics of interest from Aerospace & Electronic Systems, to Entrepreneur Network to Women in Engineering to Young Professionals. The chapters and affinity groups organize more than 100 meetings a year. In addition to the IEEE organization activities, the Boston Section organizes and sponsors up to seven conferences in any given year, as well as more than 45 short courses. The Boston Section publishes a bi-weekly newsletter and, currently, a monthly Digital Reflector newspaper included in IEEE membership.
The IEEE Boston Section also offers social programs such as the section annual meeting, Milestone events, and other non-technical professional activities to round out the local events. The Section also hosts one of the largest and longest running entrepreneurial support groups in IEEE.
More than 150 volunteers help create and coordinate events throughout the year.